TL;DR:
- Effective lab logistics are critical for maintaining reagent viability, regulatory compliance, and research integrity. Structured, digital, and validated practices in cold chain management, risk mitigation, and EU-specific regulations enable reliable supply chains and foster scientific credibility. Investing in digital tools, resilient sourcing, and strategic governance transforms logistics from an operational necessity into a competitive advantage.
Laboratory managers and procurement specialists often treat logistics as a back-office function, relegated to tracking shipments and negotiating freight rates. That perception is a costly error. Effective lab logistics actively determines whether temperature-sensitive reagents arrive viable, whether cold chain compliance holds under GDP requirements, and whether research institutions can defend the integrity of their results under regulatory scrutiny. This article covers modern logistics practices, digital tools, risk mitigation strategies, and EU-specific compliance considerations—all structured to equip decision-makers with actionable frameworks for a more reliable supply chain.
Table of Contents
- Why logistics matters in the laboratory supply chain
- Best-in-class logistics practices for lab reagents and bacteriostatic water
- Managing risk and disruption in lab supply chains
- Smart inventory management and digital tools for logistics efficiency
- Global nuances: Meeting regulatory and EU-specific logistics challenges
- A new mindset: Why logistics is your lab’s strategic differentiator
- Take your next step: Labware and reagent solutions for a resilient supply chain
- Frequently asked questions
Key Takeaways
| Point | Details |
|---|---|
| Logistics safeguards quality | Proper logistics is essential for preserving the integrity and compliance of lab reagents and bacteriostatic water. |
| Advanced tracking reduces risk | Real-time visibility, validated packaging, and monitoring are crucial for safe transport. |
| Diversification builds resilience | Dual sourcing and geographic diversification protect labs from global supply disruptions. |
| Digital tools drive efficiency | LIMS, barcode tracking, and AI reduce errors and streamline supply chain operations. |
| Compliance is complex in the EU | Efficient logistics solutions require mastering cross-border rules, serialization, and GMP in the European market. |
Why logistics matters in the laboratory supply chain
General-purpose logistics optimizes for speed and cost. Laboratory logistics operates under an entirely different set of constraints, where a two-degree temperature excursion or a missing Certificate of Analysis (CoA) can invalidate months of experimental work. The difference is not merely operational; it is scientific.
For sensitive materials such as lyophilized peptide reagents, sterile diluents, and bacteriostatic water, the supply chain is an extension of the laboratory itself. Every node in that chain, from manufacturer to courier to receiving dock, must maintain conditions that preserve biological and chemical stability. This requires structured governance across four core functions:
- Cold chain management: Maintaining validated temperature ranges (typically 2°C to 8°C for most reagents) from dispatch to delivery, including qualified packaging systems and excursion response protocols.
- Real-time tracking: Continuous monitoring of location and environmental conditions, using data loggers and IoT-enabled sensors that create an auditable temperature record.
- Documentation and traceability: GDP-compliant paperwork, including batch certificates, chain-of-custody records, and delivery confirmations, all maintained to satisfy regulatory inspections.
- Validated packaging: Selection and qualification of primary and secondary packaging materials tested for the specific lane, transit duration, and ambient temperature ranges of each route.
“Laboratory logistics is not just about moving products—it is about protecting scientific outcomes. Compliance failures at any logistics node create ripple effects that compromise research validity and regulatory standing.”
Building reliable reagent supply chains requires treating these four functions as interdependent systems, not isolated tasks. A CoA that arrives after the reagent has been consumed, or temperature data that shows a gap in monitoring, creates regulatory exposure that no amount of post-hoc documentation can fully remediate. The financial and scientific costs of logistics failure routinely exceed the cost of implementing best practices.
Best-in-class logistics practices for lab reagents and bacteriostatic water
The gap between standard logistics and best-in-class lab logistics is most visible in the level of validation applied at each step. Traditional cold chain methods rely on passive temperature packaging and periodic spot-checks, whereas digitally optimized approaches use continuous monitoring, lane qualification studies, and validated packaging systems specific to each transport corridor.
Traditional vs. digitally optimized cold chain methods
| Feature | Traditional cold chain | Digitally optimized cold chain |
|---|---|---|
| Temperature monitoring | Periodic manual checks | Continuous IoT data loggers |
| Packaging validation | Generic insulated shipper | Lane-specific qualification studies |
| Excursion response | Reactive, post-delivery review | Real-time alerts, in-transit intervention |
| Documentation | Paper-based, batch reconciled | Digital, auto-generated audit trails |
| Visibility | Carrier milestone updates | GPS and sensor data with full transparency |
| Compliance evidence | Manual records | Automated regulatory-ready reports |
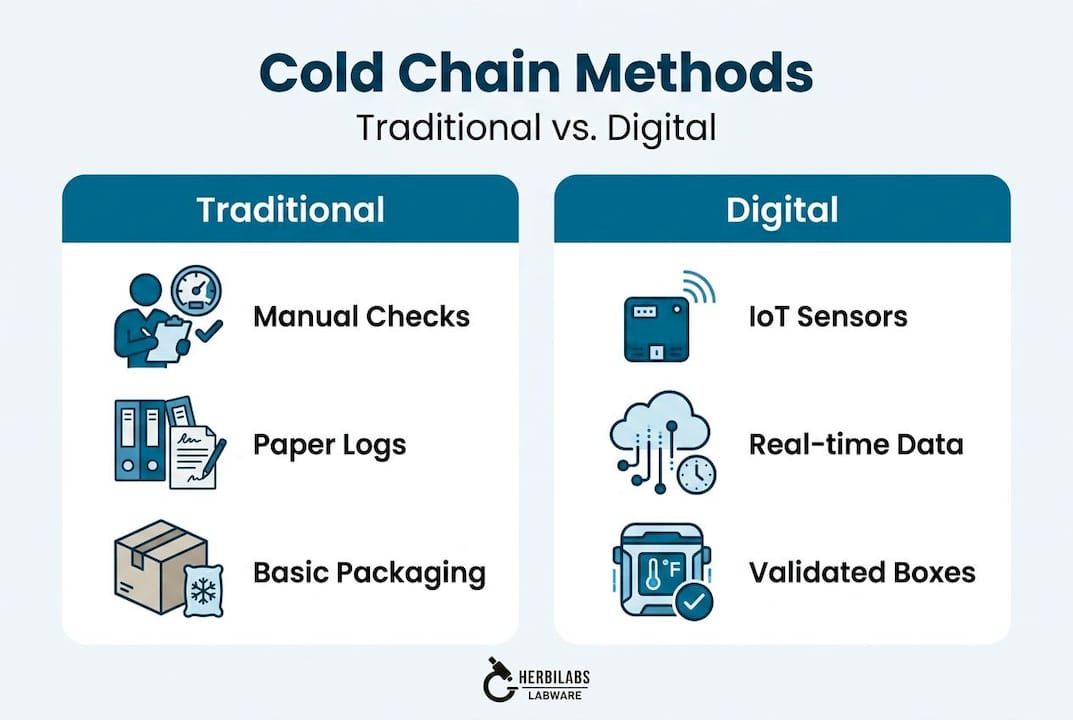
Key methodologies that separate leading logistics providers from standard carriers include lane qualification and IoT monitoring, which together ensure that every shipment of reagents or bacteriostatic water travels within a validated, continuously recorded envelope of conditions.
Sequential steps for validated reagent shipment
- Risk assessment and lane qualification: Map all transit routes, identify worst-case seasonal temperature profiles, and conduct thermal studies to validate packaging performance across those lanes.
- Packaging selection and qualification: Select packaging systems validated for the specific temperature range, transit duration, and packing configuration required for each product class.
- Pre-shipment quality verification: Confirm batch documentation, CoA, and expiration dates are complete before dispatch. Flag any discrepancies before materials leave the facility.
- Data logger calibration and activation: Calibrate and activate data loggers inside every shipment to generate a continuous, time-stamped temperature record throughout transit.
- Real-time monitoring during transit: Configure IoT alerts to notify logistics coordinators immediately if temperature thresholds are breached, allowing for in-transit intervention where feasible.
- Delivery confirmation and record archiving: Obtain signed delivery confirmation, download data logger records, and archive all documentation against the batch number for traceability and future audit.
Reviewing safe reagent handling tips alongside these shipping steps ensures that receiving protocols at the laboratory end align with the standards maintained during transit. Similarly, integrating validated shipment procedures with aseptic reagent preparation protocols reduces the risk of contamination events occurring between receipt and use.
Pro Tip: Conduct a transport risk assessment at least annually, or whenever a new lane is introduced. Packaging materials that passed qualification under winter conditions may fail in summer heat, and lane-specific performance data should drive packaging updates rather than assumptions based on product category alone. A well-maintained labware checklist that includes packaging qualification schedules helps teams stay ahead of these review cycles.
Managing risk and disruption in lab supply chains
Even a well-designed logistics framework is vulnerable to supply chain shocks: raw material shortages, single-source dependencies, geopolitical disruptions, and port congestion have all demonstrated their capacity to halt research programs. The response is not reactive improvisation but structured resilience planning embedded in procurement strategy.
Dual sourcing and geographic diversification, combined with a maintained register of critical materials, form the foundation of any robust risk mitigation program.
Risk mitigation approaches and their advantages
| Strategy | Implementation | Key advantage |
|---|---|---|
| Dual sourcing | Qualify two or more suppliers per critical reagent | Eliminates single-source dependency |
| Critical materials register | Maintain a live register of high-impact, low-substitutability items | Enables priority-based contingency planning |
| Geographic diversification | Source from suppliers in different regions or countries | Reduces exposure to regional disruption events |
| Safety stock thresholds | Define minimum inventory levels for critical reagents | Buffers against short-term supply shocks |
| Supplier qualification audits | Regularly audit supplier capabilities and continuity plans | Confirms readiness before disruptions occur |
A common misconception is that local distributors provide insulation from global supply constraints. They do not. Local distributors typically draw from the same global manufacturing base as larger direct suppliers. When a single API manufacturer faces capacity issues or a freight corridor closes, the disruption propagates equally to local and international distributors. Procurement teams must map their supply chains at least two tiers deep to identify hidden single points of failure.
“The cost of maintaining a safety stock or a qualified backup supplier is almost always lower than the cost of a research delay caused by a stockout. Labs that quantify this trade-off are far better positioned to justify resilience investments to institutional leadership.”
Practical steps for building supply chain resilience in a laboratory setting include the following:
- Register all critical reagents and bacteriostatic water products with their primary and backup suppliers in a centralized procurement database.
- Map the geographic origin of each critical raw material to identify regional concentration risk.
- Define and communicate par levels (minimum stock thresholds) to both procurement and laboratory staff, so that reorder triggers are consistent and proactive.
- Conduct annual scenario planning exercises that simulate a 4-week to 8-week supply disruption for each critical reagent category.
- Review laboratory supply chain strategies periodically and update supplier qualification status based on audit results and market intelligence.
Integrating these steps with a focus on streamlining lab workflow ensures that risk mitigation does not create operational friction but instead supports the efficiency and predictability that research programs require.
Smart inventory management and digital tools for logistics efficiency
Inventory management is the connective tissue between procurement strategy and day-to-day laboratory operations. Without structured systems, reagent expiry events, unexpected stockouts, and compliance documentation gaps accumulate silently until they cause a critical failure.

Modern laboratory inventory management applies a combination of well-established principles and emerging digital technologies to maximize efficiency, reduce waste, and maintain traceability. FIFO rotation, par levels, and LIMS integration together form a robust foundation for reagent stock control.
Steps to implement effective lab inventory management
- Apply FIFO (first in, first out) rotation: Organize storage so that the oldest batches of each reagent are used before newer stock, reducing expiry-related waste and maintaining consistent experimental conditions.
- Define and document par levels: Establish minimum and maximum stock quantities for every critical reagent based on consumption rates, lead times, and safety stock requirements. Review these quarterly.
- Implement LIMS or inventory software: Deploy a Laboratory Information Management System (LIMS) or dedicated inventory platform that tracks stock levels, batch numbers, expiration dates, and CoA documents in a single, searchable database.
- Integrate barcode or RFID scanning: Assign barcodes or RFID tags to all reagent containers so that receipts, transfers, and dispensing events are recorded automatically, reducing manual data entry errors.
- Configure expiration and reorder alerts: Set automated alerts within the LIMS so that procurement is notified when stock approaches par level thresholds or when expiration dates fall within a defined warning window.
- Conduct regular physical audits: Cross-reference physical stock counts against LIMS records at scheduled intervals to identify discrepancies, detect losses, and confirm that data integrity is maintained.
The potential impact of digital transformation in this space is significant. AI adoption in pharma logistics currently shows 36% of organizations in experimental use and 47% with AI on their roadmap; benchmarks from general supply chain implementations indicate a 15% reduction in logistics costs and a 35% improvement in inventory accuracy for early adopters. These figures suggest that labs investing in digital inventory infrastructure now will hold a measurable advantage in cost control and compliance readiness within the next three to five years.
Pro Tip: Connect your digital inventory alert system directly to your procurement scheduling calendar. When a reorder alert fires, it should automatically populate a procurement task with the relevant supplier details, par levels, and lead times. This eliminates the informal communication gap where alerts are seen but not acted upon promptly, which is the most common cause of near-miss stockout events.
Achieving these gains requires balancing automation with informed human oversight. Automated alerts and AI-driven demand forecasting improve speed and consistency, but a qualified procurement specialist reviewing lab workflow efficiency data regularly ensures that system parameters remain aligned with actual research demand patterns.
Global nuances: Meeting regulatory and EU-specific logistics challenges
The European laboratory supply chain operates across a uniquely complex regulatory landscape. With 27 member states, each maintaining national transposition of EU directives alongside country-specific licensing requirements for importation, storage, and distribution of research-grade materials, procurement teams face a level of regulatory heterogeneity that is unmatched in most other global markets.
Centralized GMP customization, serialization, and regulatory licensing across multiple EU markets are the primary operational tactics used by leading distributors to manage this complexity efficiently.
Key tactical approaches for procurement specialists operating across EU markets include:
- Centralize compliance process ownership: Designate a dedicated compliance function responsible for tracking regulatory changes across all active procurement markets. Decentralizing this responsibility across individual laboratory managers creates inconsistency and increases the risk of non-compliance.
- Implement serialization for traceability: Ensure that all reagents and reconstitution solutions entering the supply chain carry unique serialization identifiers compliant with the EU’s Falsified Medicines Directive (FMD) or equivalent national frameworks, enabling end-to-end traceability.
- Adapt documentation for each jurisdiction: While core GMP documentation is standardized, import licenses, storage authorizations, and controlled substance schedules vary by country. Maintain jurisdiction-specific documentation sets for every market where materials are procured or delivered.
- Build regional distribution buffer stock: Positioning certified safety stock at strategically located regional distribution centers within the EU reduces cross-border transit frequency and associated customs risk.
- Conduct regular regulatory horizon scanning: EU regulatory amendments, such as updates to Annex 15 validation requirements or country-specific GDP inspection findings, can alter logistics requirements with relatively short notice. Structured horizon scanning prevents reactive scrambling.
For institutions procuring across multiple EU countries simultaneously, the cumulative burden of these requirements argues strongly for partnering with logistics providers that have demonstrated, documented competency in EU-wide distribution rather than relying on ad hoc arrangements with national carriers.
A new mindset: Why logistics is your lab’s strategic differentiator
The conventional view positions logistics as a cost center to be minimized. That framing is not only outdated—it is actively counterproductive for research institutions that compete for funding, talent, and regulatory standing.
The emergence of decentralized clinical trials (DCTs) offers a compelling illustration. DCTs require reagents and biological samples to move between patient locations, satellite collection sites, and central laboratories with the same rigor previously reserved for controlled clinical facilities. This model demands logistics frameworks that are not just compliant but genuinely adaptive, capable of scaling up or down based on trial enrollment patterns and geographic expansion. Labs that have built flexible, data-driven logistics infrastructure are positioned to participate in this growing segment. Those still relying on manual inventory tracking and reactive supplier management are not.
The next significant shift will be the adoption of digital twins in laboratory supply chain management. A digital twin creates a real-time virtual representation of the entire supply chain, allowing managers to simulate disruptions, test alternative sourcing strategies, and optimize inventory levels before committing to operational changes. Early adopters in pharmaceutical logistics are already using this technology to reduce scenario planning time from weeks to hours. Research institutions that wait for widespread adoption before building the necessary data infrastructure will find themselves years behind when the capability becomes a procurement standard.
Perhaps most importantly, proactive logistics governance changes how institutional stakeholders perceive laboratory operations. Labs that can demonstrate GDP compliance, full traceability, and documented risk mitigation attract more favorable audit outcomes, which in turn strengthens their position when seeking grant funding or regulatory approval for novel research programs. Maintaining labware integrity strategies as a visible priority signals to both regulators and funding bodies that the institution operates at the highest standards of scientific rigor.
Laboratory managers who understand this dynamic have both the evidence and the framing to advocate for logistics investment at the C-suite level. Logistics is not a service cost—it is infrastructure for scientific credibility.
Take your next step: Labware and reagent solutions for a resilient supply chain
The practices described throughout this article require not only sound internal processes but also suppliers whose quality and logistics standards match the rigor of your research environment.

Herbilabs supports procurement specialists and laboratory managers across Europe by supplying high-purity reconstitution solutions manufactured to strict research-grade standards. Before your next procurement cycle, review the labware purity guide to benchmark your current product selection against best-practice purity and documentation standards. For a structured review of how your institution’s sourcing strategy aligns with resilient supply chain principles, the supply chain guidance resource provides an actionable starting point. Herbilabs also offers wholesale pricing and dedicated account support for institutional customers managing recurring procurement requirements.
Frequently asked questions
What logistics practices keep lab reagents and bacteriostatic water safe during transit?
Continuous temperature monitoring, validated packaging, and IoT visibility are the core practices that maintain reagent and bacteriostatic water integrity from dispatch through delivery confirmation.
How do labs prevent supply chain disruptions for critical reagents?
Dual sourcing and geographic diversification, paired with a maintained critical materials register, are the most effective structural defenses against supply disruptions for critical laboratory reagents.
What digital tools have the biggest impact on lab supply chains?
LIMS software, barcode tracking, and AI-powered inventory systems deliver the largest measurable improvements in cost control, expiry management, and documentation accuracy across laboratory supply chains.
What are the biggest logistics challenges for EU labs?
Regulatory variation across 27 markets, combined with serialization requirements and jurisdiction-specific licensing for research materials, represents the primary logistics complexity facing procurement teams operating across the European Union.



