TL;DR:
- Clear terminology distinguishes between lab consumables and labware, reducing ordering errors and workflow disruptions.
- Industry standards like ANSI/SLAS and ISO ensure compatibility, accuracy, and safety in laboratory consumables.
- Maintaining a standardized lexicon and documentation improves procurement precision and research reliability.
Confusion over the precise meaning of terms like “consumables,” “labware,” “pipette tip,” or “reservoir” is more common in laboratory settings than most procurement teams acknowledge, and the consequences can be significant. A single mislabeled purchase order referencing “PCR tubes” when the protocol requires “PCR strips” can halt a workflow for days, waste budget, and compromise experimental integrity. This guide systematically clarifies the core terminology surrounding lab consumables and labware, providing researchers, lab managers, and procurement specialists with a reliable reference that supports accurate ordering, consistent documentation, and regulatory compliance.
Table of Contents
- Distinguishing lab consumables and labware: Why definitions matter
- Key terms and categories: A practical glossary for lab teams
- Industry standards and compliance: What every lab should know
- Choosing the right consumables: Practical tips for reliability and efficiency
- Why clear lab consumables terminology drives research success
- Explore high-quality lab consumables for every research need
- Frequently asked questions
Key Takeaways
| Point | Details |
|---|---|
| Clear definitions prevent errors | Using precise terminology separates consumables from labware and avoids costly mistakes. |
| Industry standards matter | Following recognized standards ensures lab supplies are reliable and automation-ready. |
| Documentation streamlines procurement | Matching supplier specs with internal terms simplifies ordering, management, and compliance. |
| Growing market, evolving terms | As demand and innovation increase, labs must stay updated on evolving consumables language. |
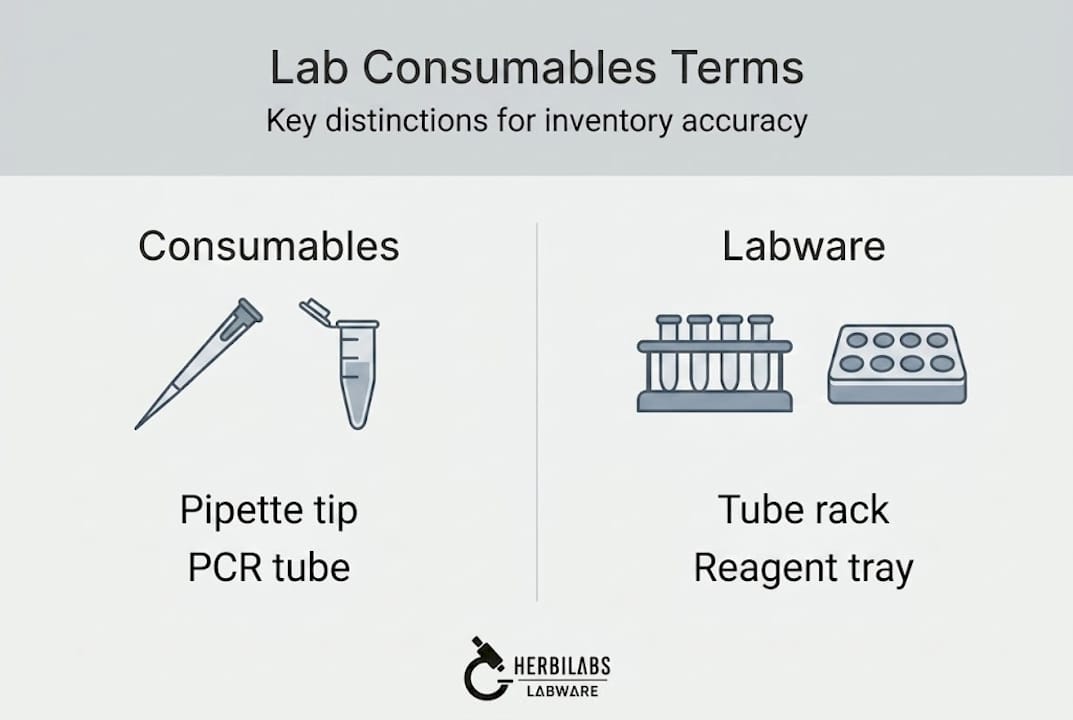
Distinguishing lab consumables and labware: Why definitions matter
Now that you’ve seen why terminology confusion is a real issue, let’s start by breaking down two key terms that often get mixed up.
The term lab consumables refers to a well-defined category of items. Laboratory consumables are expendable, single-use or limited-use items deployed in lab workflows, discarded after use, and essential for maintaining sterility, preventing cross-contamination, and ensuring accurate results. Examples include pipette tips, microcentrifuge tubes, PCR tubes, syringe filters, and disposable transfer pipettes. Because these items contact samples directly and are discarded after each use or after a defined number of uses, their material composition and certification status carry direct implications for data quality.
Labware, by contrast, covers a broader set of physical components. Labware refers to physical components such as plates, tubes, racks, and reservoirs that interface with robotic systems or manual workflows, and may be either disposable or reusable, engineered for precision and durability. The distinction matters because labware selection criteria include mechanical robustness, dimensional tolerances, and compatibility with automated liquid handling platforms, while consumables selection focuses more on sterility, lot-to-lot consistency, and material purity.
Our essential consumables guide explores how these categories interact in daily research workflows.
“Mislabeling a consumable as labware, or vice versa, in a procurement system can route the item to the wrong budget code, the wrong storage location, and the wrong quality control process, disrupting workflows at multiple points simultaneously.”
Comparison: Lab consumables vs. labware
| Feature | Lab consumables | Labware |
|---|---|---|
| Typical use duration | Single use or limited use | Reusable or disposable |
| Primary selection criteria | Sterility, purity, lot consistency | Dimensional tolerance, durability |
| Material examples | Polypropylene, nylon, PVDF | Polycarbonate, borosilicate glass, stainless steel |
| Automation compatibility | Often format-specific | Engineered to SBS/ANSI standards |
| Budget category | Operating expenditure | Capital or operating expenditure |
Common consumables: pipette tips, PCR tubes, microcentrifuge tubes, syringe filters, cell culture flasks, serological pipettes, reagent reservoirs (single-use).

Common labware: microplates, tube racks, tip boxes, reagent reservoirs (reusable), pipette stands, wash basins.
Consistent use of these definitions across procurement, storage, and protocol documentation prevents misrouting of orders and reduces the risk of using an incorrect item class in a critical assay. Our labware checklist provides a structured reference for verifying that your inventory aligns with these definitions.
Key terms and categories: A practical glossary for lab teams
With the foundation laid, let’s make terminology even clearer by detailing key terms and categories you’ll encounter in every lab.
Labware refers to components such as plates, reservoirs, tip boxes, tubes, and racks, engineered for both manual and robotic use. Understanding the specific vocabulary for each item type allows lab teams to communicate precisely with suppliers, write unambiguous protocols, and train new staff efficiently.
Essential glossary: Core lab consumables and labware terms
| Term | Category | Definition |
|---|---|---|
| Pipette tip | Consumable | Disposable plastic attachment for micropipettes; available in filtered and non-filtered variants |
| Microplate | Labware/consumable | Multi-well plate (typically 96, 384, or 1536 wells) used for assays, cell culture, or storage |
| PCR tube | Consumable | Thin-walled polypropylene tube designed for thermal cycling applications |
| Syringe filter | Consumable | Membrane-based device attached to a syringe for sterile filtration of solutions |
| Reagent bottle | Labware | Glass or plastic container for storing chemical reagents; may be reusable |
| Reservoir | Labware/consumable | Trough-shaped vessel used to hold reagents during multichannel pipetting; available in single-use and reusable formats |
| Rack | Labware | Structural holder for tubes, plates, or tips; typically reusable |
| Vial | Consumable/labware | Small sealed container for storing samples or reagents; glass or plastic |
| Tip box | Labware | Rigid container housing pipette tips; the box is labware, the tips inside are consumables |
| Filter plate | Consumable | Microplate with filter membranes in each well for filtration-based assays |
This distinction between the container (labware) and its contents or accessories (consumables) is a frequent source of ordering errors. A procurement specialist requesting “tip boxes” may receive only the boxes without tips, or vice versa, depending on how the supplier interprets the order.
Categories by use frequency:
- Single-use items: pipette tips, PCR tubes, syringe filters, cell culture flasks, serological pipettes
- Limited-use items: microplates (can be washed and reused in non-sterile applications), reagent reservoirs (reusable versions rated for a defined number of autoclave cycles)
- Reusable items: racks, tip boxes, reagent bottles, wash basins, pipette stands
Understanding these use-frequency categories directly affects stock management. Single-use items require higher reorder frequency and tighter inventory tracking, while reusable labware requires maintenance logs and periodic inspection for wear or contamination.
Pro Tip: When placing orders, always match the terminology in your purchase order exactly to the supplier’s catalog description, including format (e.g., “low-retention, filtered, 200 µL pipette tips, racked”) to prevent costly misorders and delays.
A practical illustration: a research team at a university genomics facility once ordered “PCR plates” without specifying skirt type. The supplier shipped full-skirted plates, while the team’s thermal cycler required semi-skirted plates. The resulting incompatibility caused a two-day delay while the correct format was sourced. A single additional descriptor in the original order would have prevented the issue entirely. For a structured overview of top consumables examples, refer to our dedicated resource.
Industry standards and compliance: What every lab should know
Understanding what the terms mean is essential, but industry standards bring even more clarity and reliability to the table.
Standardized terminology does not exist in isolation. It is codified and enforced through international standards that govern the design, manufacture, and use of lab consumables and labware. Automation-friendly consumables must adhere to standards including ANSI/SLAS SBS microplate specifications, ISO 8655 for volumetric systems, ISO 13485 for sterile manufacturing quality management, and USP Class VI for biocompatibility of plastics in contact with biological materials.
Each of these standards carries specific implications for terminology and procurement:
- ANSI/SLAS SBS: Defines the physical dimensions of microplates and related labware to ensure interoperability with automated liquid handling systems. A plate described as “SBS-compliant” communicates precise dimensional tolerances without ambiguity.
- ISO 8655: Applies to piston-operated volumetric apparatus, including pipettes and their tips. Compliance ensures that volumetric accuracy claims are verifiable and comparable across suppliers.
- ISO 13485: A quality management standard for medical device manufacturing, frequently applied to consumables used in clinical or regulated research environments.
- USP Class VI: Certifies that plastic materials meet biocompatibility requirements, critical for consumables used in cell-based assays or in contact with injectable preparations.
Our article on aseptic manufacturing standards provides further context on how these standards apply in sterile manufacturing environments.
Consequences of using non-compliant consumables in automated settings:
- Dimensional non-conformance causing robotic arm misalignment and plate crashes
- Material leaching from non-certified plastics contaminating samples and invalidating assay results
- Volumetric inaccuracy from non-ISO 8655 tips leading to systematic pipetting errors
- Regulatory audit failures when documentation lacks compliance certificates
- Cross-contamination events due to inadequate sterility specifications on single-use items
The global lab consumables market reflects the growing importance of these standards. The consumables market is growing at a CAGR of 10.01%, projected to reach $25.32 billion by 2033, driven substantially by biotech and pharmaceutical demand for certified, traceable consumables. This growth is inseparable from the parallel tightening of compliance requirements across regulated industries.
Pro Tip: Always request a certificate of conformance or compliance documentation from your supplier before introducing a new consumable into a regulated or automated workflow. Verify that the certificate references the specific lot number, not just the product line. Our lab quality control resource outlines what documentation to request and how to evaluate it.
Choosing the right consumables: Practical tips for reliability and efficiency
Guided by standards, your consumables selections become even more critical. Here’s how to ensure you get it right every time.
Correct terminology is not only a communication tool but also a decision-making framework. When a lab manager uses precise language to specify a consumable, every downstream process benefits: the purchasing team orders the correct item, the receiving team stores it correctly, and the researcher uses it with confidence.
Key factors for selecting lab consumables:
- Compatibility: Confirm dimensional and material compatibility with your instruments, particularly for automated platforms
- Certification: Verify that the item carries relevant compliance marks (ISO, USP Class VI, DNase/RNase-free, pyrogen-free) appropriate to your application
- Storage requirements: Some consumables (e.g., sterile filtered vials, low-binding tubes) require specific storage conditions that affect inventory planning
- User feedback and lot consistency: Review internal records or supplier data on lot-to-lot variability, particularly for critical assays
- Supply chain reliability: Assess lead times, minimum order quantities, and supplier track record to avoid stockouts during active research phases
A well-documented example of terminology-driven failure involves skirtless PCR plates in automation. Non-SBS labware may work manually but fail in automation due to mechanical robustness issues, with flimsy skirtless PCR plates being a cited example. When a lab transitions from manual to automated PCR workflows without updating its consumables specification language, the result can be plate warping, robotic misreads, and aborted runs. The fix requires not just replacing the plates but also updating the terminology in the protocol and the procurement system to prevent recurrence.
Borosilicate glassware presents a different terminology challenge. Borosilicate glass accounts for 65% of glassware demand, valued for its chemical resistance and thermal stability. However, borosilicate glassware carries a breakage rate of 10 to 15% annually, which must be factored into ordering frequency and safety protocols. When procurement records use the generic term “glass bottle” rather than “borosilicate reagent bottle,” reorder decisions may default to cheaper soda-lime alternatives that are less chemically resistant, introducing a subtle but meaningful risk to reagent integrity.
Our consumables comparison resource and quality control tips page provide structured frameworks for evaluating these factors systematically.
Pro Tip: Maintain a dual-reference log for every consumable in your inventory, recording both the supplier’s catalog part number and your lab’s internal terminology. This practice eliminates ambiguity during reorders and simplifies onboarding for new team members.
As the market grows and new product categories emerge, naming conventions will continue to evolve. Labs that establish rigorous internal lexicons today will be better positioned to adopt new terminology without confusion or procurement disruption.
Why clear lab consumables terminology drives research success
Having explored actionable best practices, let’s consider why terminology matters more than you might think, and how it functions as the unsung driver of reliable research.
In our experience working with research institutions and procurement teams, the most consequential lab errors rarely originate from instrument malfunction or reagent failure. They originate from language. A shipment of “sterile vials” that turns out to be non-sterile glass vials because the supplier’s definition of “sterile” differed from the lab’s expectation can expose samples to contamination, invalidate weeks of work, and trigger a safety review. The root cause is not negligence but terminological ambiguity.
The best-performing labs we observe treat terminology as infrastructure. They maintain a standardized internal lexicon, updated regularly, that maps every consumable and labware item to a precise definition, a supplier part number, and a compliance category. New staff are trained on this lexicon during onboarding, not as an afterthought but as a foundational competency. This approach reduces procurement errors, accelerates troubleshooting, and makes audit preparation significantly less burdensome.
There is also a safety dimension that is often underestimated. Incorrect consumables used in automated systems can cause mechanical failures that damage expensive equipment and, in some cases, create hazardous situations involving pressurized liquids or biological materials. The language used to specify consumables is the first line of defense against these outcomes.
We recommend instituting a formal “terminology check” as a standard step in both the onboarding process and the consumables ordering workflow. This check should verify that the term used in the protocol matches the term in the procurement system, which in turn matches the supplier’s catalog description. Our guide to reliable consumables provides a practical starting point for building this kind of structured approach within your lab.
Explore high-quality lab consumables for every research need
Ready to put this clarity to work in your own lab? Browse trusted suppliers that handle the details so you can focus on science.
Herbilabs offers a curated range of research-grade consumables and labware, each described with precise terminology and supported by compliance documentation. Whether you are sourcing bacteriostatic water, sterile diluents, or reconstitution solutions for peptide research, every product listing includes material specifications, lot traceability, and relevant certifications to eliminate ambiguity at the point of purchase.

Our reliable labware solutions page and top lab consumables comparison resource are designed to help procurement specialists and lab managers make fast, informed selections without navigating vague or inconsistent product descriptions. Partnering with a supplier who prioritizes terminological precision and regulatory compliance means fewer procurement errors, faster workflows, and research results you can trust.
Frequently asked questions
What is the main difference between lab consumables and labware?
Lab consumables are expendable, single-use or limited-use items discarded after use, while labware encompasses both disposable and reusable equipment designed for repeated or structural use in experiments.
Why are lab consumables standards like ANSI/SLAS and ISO important?
Automation-friendly consumables must adhere to standards such as ANSI/SLAS SBS, ISO 8655, ISO 13485, and USP Class VI to ensure dimensional compatibility, sterility, volumetric accuracy, and biocompatibility across regulated and automated environments.
What materials are most popular for lab glassware and why?
Borosilicate glass accounts for 65% of glassware demand due to its superior chemical resistance and thermal stability, making it the preferred material for reagent storage and high-temperature applications despite a 10 to 15% annual breakage rate.
How can labs avoid errors when ordering consumables?
Always align the terminology in your purchase order with the supplier’s exact catalog description, including format, material, and certification details, and maintain a dual-reference log pairing internal lab terms with supplier part numbers.
How is the lab consumables market changing?
The consumables market is projected to reach $25.32 billion by 2033 at a CAGR of 10.01%, driven by expanding biotech and pharmaceutical demand and increasingly stringent compliance requirements that are reshaping product naming conventions and certification standards.



