You trust your labware to be clean, sterile, and inert. Yet contaminants leaching from labware introduce uncontrolled variables that compromise experimental accuracy and reproducibility in sensitive assays, particularly in peptide research. Even trace plasticizers, glass-derived ions, or surface-bound residues can skew quantification, trigger false biological signals, and undermine months of work. This guide reveals how seemingly invisible impurities disrupt peptide experiments, which labware materials pose the greatest risks, and how to safeguard your results through informed material selection and validation protocols.
Table of Contents
- Why labware purity matters: Setting the stage
- How contaminants and adsorption disrupt peptide experiments
- Key labware materials: Risks and recommendations
- Automation, scale-up, and the compounding effects of impurity
- Cost versus value: Is premium labware worth it?
- Best practices for contaminant-free peptide research
- Enhance peptide research outcomes with Herbilabs Labware solutions
- Frequently asked questions
Key Takeaways
| Point | Details |
|---|---|
| Invisible risks exist | Even trace contaminants in labware can disrupt sensitive peptide experiments and skew data. |
| Material choice is critical | Polypropylene and certified low-binding plastics outperform glass or standard plastics for peptide work. |
| Scale amplifies problems | In high-throughput or automated assays, small impurity effects multiply, impacting reproducibility. |
| Investing in purity pays off | Premium labware reduces experiment failure and ensures reliable results, justifying its higher cost. |
| Best practices prevent errors | Routine validation and adherence to purity certifications keep experiments contaminant-free and reproducible. |
Why labware purity matters: Setting the stage
To understand why purity cannot be taken for granted, we must first define labware-related contamination and its specific implications for peptide science. Labware purity refers to the absence of extractables, leachables, and surface-active contaminants that can migrate into your samples. Extractables are compounds released under harsh extraction conditions during manufacturing quality tests, while leachables are substances that migrate into solutions under normal use conditions. Common culprits include plasticizers such as phthalates, mold-release agents, stabilizers, and trace metals from glass or stainless steel.
Peptide research is especially vulnerable to these contaminants because peptides are small, often hydrophobic or charged molecules that interact readily with surfaces and dissolved impurities. Even a 1% contamination can trigger false positive immune responses in T-cell assays, rendering entire experimental batches unusable. The consequences extend beyond isolated failures:
- False positives and negatives: Leached estrogenic compounds activate hormone receptors, mimicking biological activity that does not exist.
- Variability in results: Batch-to-batch differences in labware manufacturing introduce uncontrolled variables, making reproducibility nearly impossible.
- Reproducibility crises: When other laboratories cannot replicate your findings due to hidden labware contamination, the credibility of your work suffers.
“The smallest trace of a plasticizer can alter receptor binding assays, leading researchers down investigative dead ends that waste time and funding.”
For laboratories working with high-purity reagents, the irony is stark: investing in ultrapure solvents and certified peptides while overlooking labware purity undermines the entire experimental foundation.
How contaminants and adsorption disrupt peptide experiments
With the basics of purity established, let’s closely examine how seemingly invisible contaminants or adsorption dramatically impact peptide assay outcomes. Contamination mechanisms fall into two broad categories: chemical leaching and physical adsorption. Chemical leaching occurs when additives used during labware manufacturing migrate into your solutions. Standard plastics contain plasticizers to improve flexibility, stabilizers to prevent degradation, and mold-release agents to facilitate production. Under typical laboratory conditions, these compounds dissolve into aqueous or organic solvents, introducing cytotoxic or hormonally active substances into your assays.
Peptide adsorption to glass and cellulose causes major quantification errors, particularly for hydrophobic or charged sequences. Glass surfaces, despite their reputation for inertness, carry negative charges that attract cationic peptides. Cellulose-based filters bind peptides through hydrogen bonding and hydrophobic interactions. The result is a systematic loss of free peptide from solution, leading to underestimation of concentrations and skewed dose-response curves.
Standard plastics leach estrogenic and cytotoxic compounds that disrupt high-sensitivity peptide work. In one documented case, polystyrene plates released enough bisphenol A to activate estrogen receptors at concentrations below detection limits of standard analytical methods. Researchers observed biological activity in negative controls, a clear indicator of labware interference.
Consequences of contamination and adsorption include:
- Quantification errors: Loss of peptide to surfaces reduces measured concentrations, requiring higher sample volumes and increasing costs.
- Non-specific binding: Leached compounds occupy receptor sites, altering apparent affinity constants and IC50 values.
- False biological signals: Cytotoxic leachables induce cell stress responses unrelated to the peptide under investigation.
Pro Tip: When working with low-volume, high-throughput systems, the surface-to-volume ratio increases dramatically, amplifying adsorption losses. Always validate new labware with a recovery experiment using a radiolabeled or fluorescently tagged peptide to quantify surface binding before committing to large-scale studies. For additional guidance, review strategies to avoid contamination mistakes and follow reagent handling safety tips to minimize risk.
Key labware materials: Risks and recommendations
Knowing the mechanisms of contamination, it’s essential to choose the right material for your research. Here’s how common labware types compare for peptide applications:
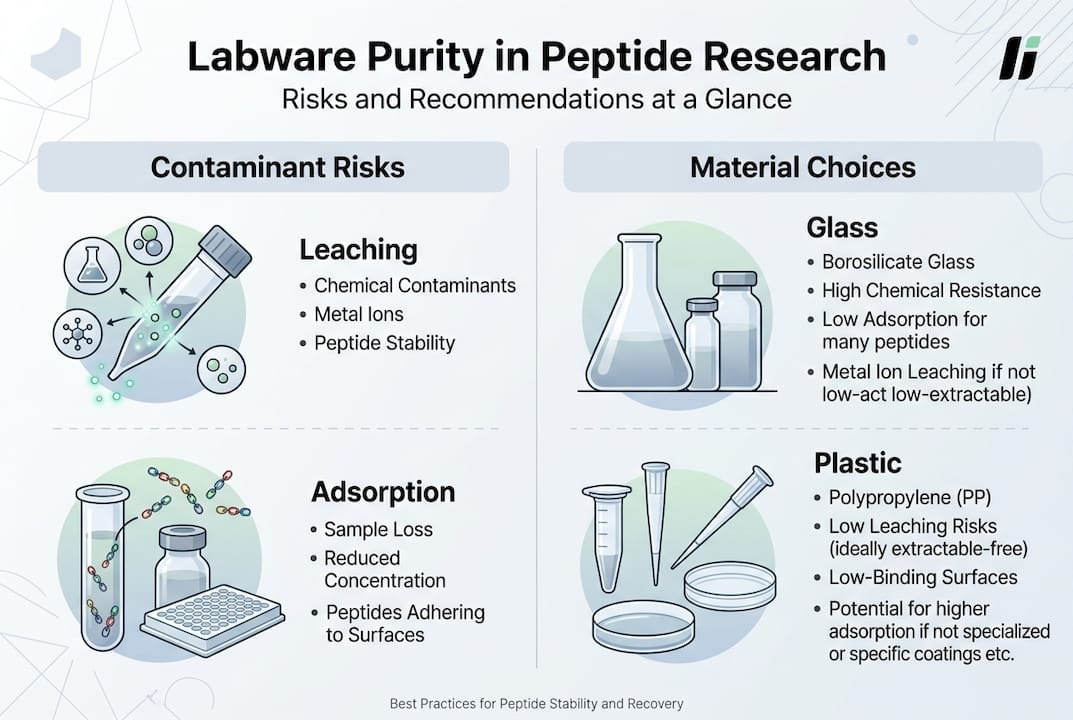
| Material | Leaching Risk | Adsorption Risk | Best Use Case | Limitations |
|---|---|---|---|---|
| Glass | Low (but metal ions possible) | High (charged peptides) | Long-term storage of non-charged peptides | Avoid for cationic or highly hydrophobic sequences |
| Standard plastic (PS, PVC) | High (plasticizers, stabilizers) | Moderate to high | General lab use, non-critical assays | Unsuitable for sensitive bioassays |
| Low-binding plastic (PP, PE) | Low to moderate | Low (surface-treated) | Peptide dilutions, assay plates | Verify certification for each batch |
| Certified virgin polymer tubes | Very low | Very low | High-sensitivity peptide quantification | Higher cost, limited suppliers |
Polypropylene and low-binding plastics are preferable for peptide handling over glass or cellulose. Surface treatments such as MaxPeak or similar proprietary coatings reduce non-specific binding by creating a hydrophilic barrier that minimizes peptide-surface interactions. When selecting labware, prioritize materials with documented low extractables profiles and certificates of analysis confirming trace metal and organic impurity levels.
For peptide-specific needs, consider these recommendations:
- Dilution and storage: Use certified low-binding polypropylene tubes with screw caps to prevent evaporation and contamination.
- Assay plates: Select surface-treated microplates designed for protein and peptide work, avoiding standard tissue culture plates.
- Filtration: Replace cellulose filters with low-protein-binding PVDF or PES membranes to minimize peptide loss.
Statistic Callout: Laboratories switching from standard glass vials to certified low-binding plastics report up to 40% improvement in peptide recovery rates, translating directly to reduced sample consumption and cost savings.
Explore lab consumables examples and implement aseptic technique essentials to maintain labware integrity throughout your workflow.
Automation, scale-up, and the compounding effects of impurity
When running peptide research at scale or via robotics, labware purity takes on even greater importance. Automated workflows process hundreds or thousands of samples sequentially, and every impurity event amplifies across the entire batch. A single contaminated tip box or microplate can introduce systematic error that invalidates an entire screening campaign. Labware purity directly impacts mass balance and scheduling accuracy in automated systems, where contamination risks scale with throughput.
Mass balance calculations depend on accurate knowledge of free peptide concentration in solution. When leachables or adsorption remove peptide from the liquid phase, the discrepancy between expected and measured concentrations grows. In high-throughput screening, this manifests as drift in assay performance over time, with early samples showing different responses than later ones due to cumulative surface saturation or leachable accumulation.
Consider the following data illustrating how small impurity levels scale in high-throughput contexts:
| Throughput Level | Samples per Run | Cumulative Leachable Exposure | Adsorption Loss per Sample | Total Error Potential |
|---|---|---|---|---|
| Low (manual) | 10-50 | Minimal | 5-10% | Manageable with controls |
| Medium (semi-automated) | 100-500 | Moderate | 5-10% per sample | Requires validation |
| High (fully automated) | 1,000-10,000 | High (cumulative buildup) | 5-10% per sample | Critical; demands certified labware |
Scheduling and throughput bottlenecks often trace back to purity problems. When assays fail quality control checks, entire plates must be repeated, consuming additional reagents, peptides, and instrument time. The hidden cost of using substandard labware includes not only failed experiments but also delayed timelines and reduced laboratory productivity.
Pro Tip: Validate automation runs with test plates using different labware brands to identify hidden leachables. Run blanks and positive controls in every batch, and monitor for trends such as increasing background signal or decreasing peptide recovery over the course of a plate. This proactive approach catches contamination before it compromises critical samples. Maintain water purity standards and ensure stability of research water to complement your labware purity efforts.
Cost versus value: Is premium labware worth it?
With technical risks explored, consider whether upfront investment in premium labware pays off for your research goals. The direct costs of experiment contamination include wasted reagents, peptides, and consumables. A single failed assay plate can consume peptides worth hundreds of euros, not to mention the labor hours spent preparing samples and running the assay. Indirect costs are even more substantial: delayed publications, missed grant deadlines, and damage to laboratory reputation when results cannot be reproduced.
Common false savings when using standard plastics include:
- Bulk purchasing of uncertified labware: Lower unit prices are offset by higher failure rates and the need to repeat experiments.
- Reusing labware not designed for reuse: Cleaning protocols may not remove all leachables, and surface degradation increases adsorption over time.
- Ignoring batch-to-batch variability: Even within a single product line, manufacturing variations introduce uncontrolled variables.
“High-purity labware reduces failure rates, despite higher upfront costs, compared to cheap plastics that may cause cytotoxic effects. The return on investment becomes clear when you calculate the cost of a single failed experiment against the marginal price difference for certified consumables.”
When evaluating labware, look for these certification indicators:
- Low-extractable claims: Manufacturer documentation showing extractables testing under relevant solvent conditions.
- Certificate of Analysis (COA): Batch-specific data on trace metals, organic impurities, and endotoxin levels.
- Regulatory compliance: ISO 13485 or similar quality management certifications indicating consistent manufacturing processes.
- Surface treatment validation: Independent testing confirming reduced protein binding for low-binding products.
For budget-conscious laboratories, a tiered approach works well: use premium certified labware for critical assays and final validation experiments, while reserving standard plastics for preliminary work and method development. This strategy balances cost control with result integrity. Review lab consumable costs to optimize your purchasing decisions.
Best practices for contaminant-free peptide research
By following a structured approach, you can protect your results and make the most of high-purity labware. Implement these steps to establish a contamination-free workflow:
- Select labware based on application-specific requirements: Match material properties to your peptide’s physicochemical characteristics. Hydrophobic peptides require low-binding surfaces, while charged peptides benefit from neutral or surface-treated plastics.
- Qualify new labware batches before use: Run recovery experiments with representative peptides to establish baseline performance. Compare new batches against previously validated lots to detect manufacturing variations.
- Implement routine validation protocols: Include blanks in every assay to monitor for leachables. Track recovery rates over time to identify trends indicating surface saturation or degradation.
- Pre-wash labware when appropriate: For glass or reusable plastics, establish validated cleaning protocols using high-purity solvents. Verify cleanliness with blank runs before introducing samples.
- Store labware properly: Keep consumables sealed until use to prevent dust and airborne contamination. Control temperature and humidity to minimize degradation of surface treatments.
- Document labware lot numbers: Maintain records linking experimental results to specific labware batches, enabling root cause analysis if contamination is suspected.
- Dispose of labware according to validated schedules: Single-use plastics should never be reused for critical assays. Establish clear guidelines for when reusable labware must be retired.
MaxPeak surfaces and low-binding labware mitigate non-specific binding and leaching, but only when used correctly. Surface treatments degrade over time, especially with repeated exposure to organic solvents or extreme pH. Always consult manufacturer guidelines for chemical compatibility and maximum reuse cycles.
Pro Tip: Test unknown batches with mock assays before use in critical experiments. Prepare a standard peptide solution at a known concentration, aliquot it into the new labware, and measure recovery after a defined incubation period. Compare results against your established baseline to confirm the labware meets your purity standards. This simple validation step prevents costly failures and builds confidence in your experimental design. Ensure proper lab safety best practices and maintain sample traceability throughout your workflow.
Enhance peptide research outcomes with Herbilabs Labware solutions
Armed with best practices, partnering with a reliable supplier is your final safeguard for contaminant-free research. Herbilabs Labware specializes in providing high-purity bacteriostatic water, sterile diluents, and reconstitution solutions manufactured to strict purity standards in dedicated facilities. Our products undergo rigorous quality control to ensure they meet the demanding requirements of peptide research, with documented low extractables profiles and batch-specific certificates of analysis.

Researchers across Europe trust Herbilabs for pure reagents for research that support experimental accuracy and reproducibility. Our commitment to quality extends beyond products to include expert guidance on safe reagent handling and contamination prevention strategies. Whether you’re scaling up peptide synthesis, validating new assay protocols, or troubleshooting unexpected results, Herbilabs provides the certified supplies and technical support you need to succeed. Explore our full range of Herbilabs Labware solutions to find the right products for your specific research applications.
Frequently asked questions
What is the biggest risk if I use standard plastic labware in peptide research?
Standard plastics leach compounds such as plasticizers and stabilizers that interfere with peptide assays, causing false positives, cytotoxic effects, and irreproducible results that undermine experimental validity.
How can I quickly test if my labware is contaminating my assays?
Run a blank control using your solvent or buffer in the labware and monitor for unexpected absorbance, fluorescence, or bioactivity signals. Routine validation and blanks detect hidden leachables before they compromise critical samples.
Does labware adsorption affect all peptides equally?
No, hydrophobic or charged peptides adsorb strongly to standard surfaces like glass and untreated plastics, leading to higher losses compared to neutral, hydrophilic sequences.
Which labware certifications best ensure purity for peptide work?
Look for certified virgin polymers, low-binding plastics, and labware with certificates of analysis documenting low extractables and trace metal levels. Certified labware with purity claims protects sensitive assays from contamination.
Can I reuse labware to reduce costs without compromising purity?
Reusing labware designed for single use introduces contamination risks and surface degradation that increase adsorption. If reuse is necessary, establish validated cleaning protocols and verify cleanliness with blank runs, but reserve new certified labware for critical experiments.



