Reagent purity isn’t just a box to check on your lab order form. It’s the invisible foundation that determines whether your experiments deliver trustworthy data or misleading noise. Many researchers assume all reagents labeled for research use meet the same standards, but purity grades vary dramatically, and those differences ripple through every stage of your work. When peptide synthesis or cell culture depends on precise molecular interactions, even trace contaminants can derail months of effort. Understanding what high-purity reagents deliver and why they matter transforms how you approach experimental design and data interpretation.
Table of Contents
- Key takeaways
- Understanding reagent purity and its impact on research
- Comparing high-purity reagents with standard reagents
- Maintaining reagent purity: storage and handling best practices
- Regulatory standards and certifications ensuring reagent purity
- Explore high-purity reagents and expert labware solutions
- Frequently asked questions about high-purity reagents
Key Takeaways
| Point | Details |
|---|---|
| Benefits of high purity | High purity reagents minimize interference from contaminants and improve data reliability and reproducibility. |
| Regulatory compliance | Regulatory bodies and journals increasingly require documented reagent purity through certificates and traceability. |
| Purity affects outcomes | Trace contaminants can alter measurements, enzyme kinetics, or cell viability, producing misleading results. |
| Common contamination sources | Common sources include manufacturing residues, storage degradation, leachables from containers, cross contamination, and environmental exposure. |
Understanding reagent purity and its impact on research
Reagent purity describes the percentage of a substance that consists of the intended compound versus impurities like residual solvents, heavy metals, endotoxins, or degradation products. Laboratories typically encounter purity grades ranging from technical (70-90% pure) to analytical (95-99%) to ultra-pure (99.5%+). The grade you choose shapes everything from baseline noise in spectroscopy to cell viability in culture systems.
Impurities act as invisible saboteurs in your protocols. A trace amount of metal ion contamination can catalyze unwanted side reactions during peptide coupling. Endotoxins at parts-per-billion levels trigger inflammatory responses in cell assays, creating false signals that mimic genuine biological activity. When results vary inexplicably between runs, contamination from reagents often explains the mystery.
High-purity reagents minimize interference from these contaminants that skew measurements and compromise reproducibility. Consider a simple pH buffer: technical-grade components might introduce ions that shift electrode readings by 0.1 pH units, enough to alter enzyme kinetics or protein stability in sensitive applications. Ultra-pure alternatives eliminate this variable entirely.
Regulatory bodies and journal editors increasingly demand documented reagent purity. ISO 17025 accredited labs must demonstrate traceability for all materials used in testing. Pharmaceutical research following Good Laboratory Practice requires certificates proving reagent specifications. Even academic publications now request detailed methods sections identifying reagent grades and suppliers.
“The purity of your reagents sets the ceiling for your data quality. You cannot measure phenomena more precisely than the materials you use to create experimental conditions.”
Common contamination sources include:
- Manufacturing residues from synthesis processes that aren’t fully removed
- Degradation products formed during storage under improper conditions
- Leachables from container materials that migrate into solutions
- Cross-contamination during handling in non-sterile environments
- Environmental exposure to dust, moisture, or reactive gases
Understanding lab water purity standards provides context for why reagent specifications matter so critically. Water serves as the universal solvent in most lab work, and its purity directly affects every solution you prepare. The same principle applies to all reagents: purity isn’t an abstract specification but a practical determinant of whether your experiments succeed or fail.
Comparing high-purity reagents with standard reagents
The gap between high-purity and standard reagents becomes clear when you examine their specifications side by side. This comparison reveals why sensitive applications demand premium materials.
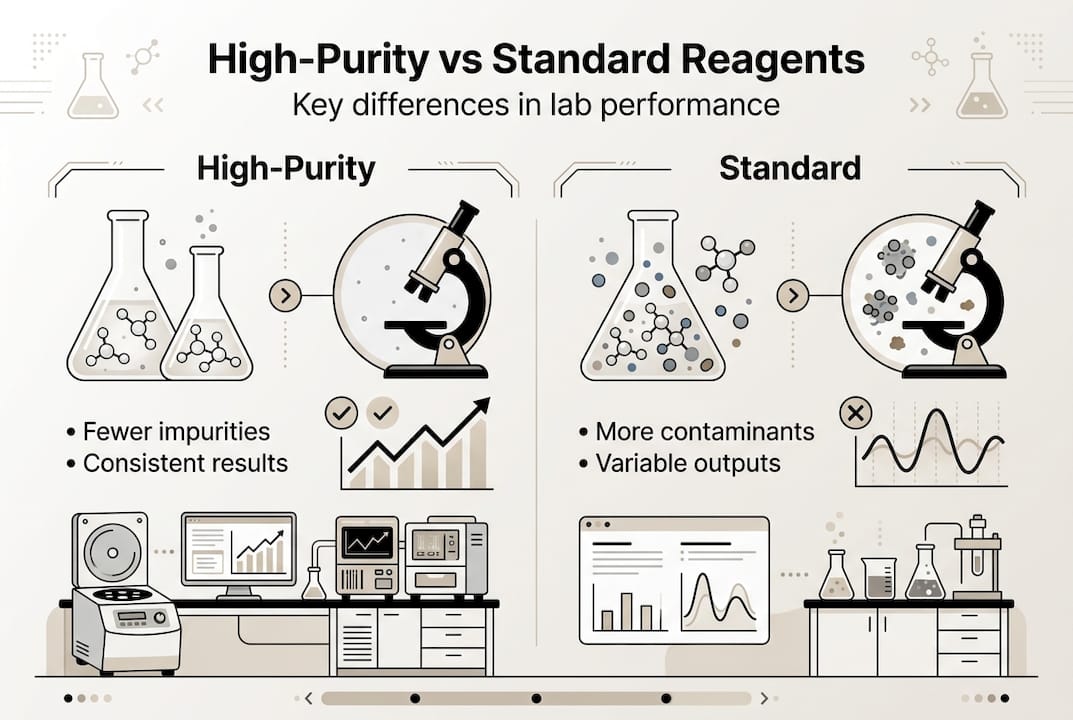
| Feature | Standard Reagents | High-Purity Reagents |
|---|---|---|
| Typical purity | 95-98% | 99.5-99.99% |
| Contaminant testing | Limited parameters | Comprehensive panel |
| Batch consistency | Variable | Tightly controlled |
| Certificate detail | Basic composition | Full analytical data |
| Cost per gram | Lower initial price | Higher upfront investment |
| Risk of interference | Moderate to high | Minimal |
| Suitable applications | Routine synthesis, general prep | Peptide work, cell culture, analytical |
Using certified high-purity reagents drastically reduces contamination and experimental variability. When you’re synthesizing peptides, that extra 1.5% purity translates to fewer truncated sequences and higher yields of your target compound. In cell culture, ultra-pure media components prevent toxic responses that standard grades might trigger.

Cost calculations must account for total experimental expense, not just reagent price per unit. A failed experiment due to contaminated materials wastes not only the reagents themselves but also researcher time, instrument hours, and opportunity cost. When a single contamination event ruins a week of work, the premium for high-purity materials becomes trivial compared to the loss.
Peptide synthesis illustrates these stakes perfectly. Each coupling step depends on precise stoichiometry and clean reaction conditions. Standard-grade coupling reagents might contain trace amines that compete with your target sequence, creating deletion peptides that contaminate your final product. High-purity alternatives eliminate these competing reactions, dramatically improving synthesis efficiency.
Selection criteria for sensitive applications include:
- Verify lot-specific certificates of analysis before purchasing
- Check testing dates to ensure recent quality verification
- Confirm storage and shipping conditions maintain integrity
- Review impurity profiles for compounds that affect your specific application
- Consider supplier reputation and quality management systems
Pro Tip: Always request and save the certificate of analysis for every reagent batch you use. When troubleshooting unexpected results months later, these documents provide crucial clues about whether reagent quality contributed to anomalies. Archive them with your lab notebooks as permanent records.
The performance difference extends beyond immediate experimental success. High-purity reagents enable you to push detection limits lower, resolve smaller differences between samples, and publish data that withstands scrutiny during peer review. They’re not luxury items but essential tools for research that aims to advance knowledge rather than simply generate activity.
Mastering aseptic reagent preparation techniques complements your investment in pure starting materials. Even ultra-pure reagents become compromised if handled carelessly, so proper technique throughout the workflow protects your quality standards from purchase through final use.
Maintaining reagent purity: storage and handling best practices
Purchasing high-purity reagents represents only the first step. Preserving that purity throughout storage and use requires systematic attention to environmental conditions and handling protocols. Degradation often occurs silently, compromising performance before visible changes alert you to problems.
Temperature control prevents thermal degradation and maintains chemical stability. Most reagents specify storage ranges like 2-8°C for refrigeration or negative 20°C for freezing. Exceeding these limits even briefly can trigger decomposition reactions. Room temperature storage suits only the most stable compounds, and even these benefit from cool, consistent conditions.
- Receive reagents promptly and transfer to proper storage immediately upon delivery
- Verify storage temperature with calibrated thermometers, not just unit displays
- Minimize freeze-thaw cycles by aliquoting reagents into single-use portions
- Seal containers tightly after each use to prevent moisture absorption
- Label all aliquots with reagent name, concentration, date, and storage requirements
- Maintain detailed logs tracking when containers are opened and environmental exposures
- Inspect reagents before each use for visible signs like color change or precipitation
- Discard materials past expiration dates regardless of appearance
Light exposure degrades photosensitive compounds through oxidation and radical formation. Amber bottles provide basic protection, but light-sensitive reagents require storage in darkness. Fluorescent lab lighting contains UV wavelengths that accelerate breakdown even through colored glass.
Proper storage conditions are critical to maintaining reagent stability and purity over time. Temperature fluctuations, humidity, and light all contribute to gradual quality loss that accumulates until reagents no longer perform as specified. Your storage practices either preserve or destroy the purity you paid for.
Container selection matters more than many researchers realize. Glass remains inert for most applications, but plastic containers can leach plasticizers, stabilizers, or monomers into solutions. High-density polyethylene and polypropylene offer better chemical resistance than standard plastics. For ultra-pure applications, pre-cleaned glass with PTFE-lined caps provides maximum protection.
Contamination during handling represents the most preventable purity loss. Every time you open a container, you expose contents to room air containing dust, microbes, and volatile compounds. Proper lab reagent storage best practices include working in clean environments, using sterile technique, and never returning unused portions to stock bottles.
Pro Tip: Create a reagent inventory system that tracks not just what you have but when containers were opened and how many freeze-thaw cycles they’ve experienced. This simple record-keeping prevents using degraded materials and helps identify patterns when unexpected results occur.
Humidity control prevents hygroscopic reagents from absorbing water, which dilutes concentrations and can trigger hydrolysis reactions. Desiccators with fresh desiccant protect opened containers between uses. Vacuum desiccators offer additional protection for the most sensitive compounds.
Environmental monitoring extends beyond just temperature. Consider factors like vibration from nearby equipment, electromagnetic fields from instruments, and chemical vapors from adjacent work areas. These subtle influences accumulate over months of storage, gradually compromising reagent integrity.
Documentation transforms storage from a passive activity into quality control. Record storage temperatures daily, note any deviations immediately, and track corrective actions. This data becomes invaluable when investigating whether storage issues contributed to experimental problems. Understanding how to approach avoiding reagent contamination systematically prevents most common purity losses.
Regulatory standards and certifications ensuring reagent purity
Regulatory frameworks establish minimum purity requirements and testing protocols that protect research quality and safety. Understanding these standards helps you evaluate supplier claims and select reagents that meet your compliance obligations.
ISO 9001 certification demonstrates that manufacturers maintain quality management systems with documented procedures, regular audits, and continuous improvement processes. This standard doesn’t specify reagent purity itself but ensures consistent manufacturing practices that deliver predictable quality.
ISO 17025 accreditation applies to testing laboratories and verifies their competence to perform specific analytical methods. When a supplier’s analytical lab holds this accreditation, you can trust that purity measurements reflect true values rather than artifacts of poor testing technique.
| Certification | What It Guarantees | Relevance to Reagent Purity |
|---|---|---|
| ISO 9001 | Quality management systems | Consistent manufacturing processes |
| ISO 17025 | Testing lab competence | Accurate analytical measurements |
| GMP (Good Manufacturing Practice) | Pharmaceutical-grade production | Strict contamination controls |
| USP (United States Pharmacopeia) | Specific purity standards | Defined limits for impurities |
| EP (European Pharmacopoeia) | European purity standards | Harmonized specifications across EU |
| Certificate of Analysis | Batch-specific test results | Actual purity of material you receive |
Certificates of Analysis provide the most direct evidence of reagent purity. These documents report actual test results for the specific lot you’re purchasing, not just general specifications. Certifications and COAs provide reliable assurance of reagent purity and compliance with regulations by documenting exactly what’s in each batch.
A comprehensive COA includes:
- Lot or batch number uniquely identifying the material
- Manufacturing and testing dates showing data currency
- Purity percentage determined by validated analytical methods
- Impurity profiles listing specific contaminants and their levels
- Physical properties like appearance, pH, and solubility
- Storage recommendations and expiration dating
- Analytical methods used with references to standard procedures
European regulations increasingly require traceability throughout the supply chain. REACH (Registration, Evaluation, Authorization, and Restriction of Chemicals) mandates safety data for substances manufactured or imported above certain volumes. While focused on safety rather than purity per se, REACH compliance indicates suppliers maintain detailed records about their materials.
Good Laboratory Practice standards apply to non-clinical safety studies and require documented evidence that reagents meet specifications. If your research supports regulatory submissions, GLP compliance becomes mandatory. Even basic research benefits from GLP principles like documented procedures and quality checks.
Pharmaceutical applications demand the highest purity grades, typically meeting USP or EP monograph specifications. These pharmacopeial standards define not just minimum purity but also specific tests for likely impurities based on synthesis routes and degradation pathways. Reagents meeting these standards cost more but provide assurance needed for regulated work.
Compliance safeguards research integrity by ensuring materials perform consistently and predictably. When every researcher uses reagents meeting defined standards, results become comparable across labs and over time. This standardization accelerates scientific progress by eliminating variables introduced by inconsistent material quality.
Reviewing certificate of analysis details before purchasing allows you to verify that specifications match your requirements. Don’t assume all suppliers interpret purity grades identically. One company’s analytical grade might differ significantly from another’s, so actual test data matters more than labels.
Explore high-purity reagents and expert labware solutions
Now that you understand why reagent purity determines experimental success, you need reliable sources for materials that meet these exacting standards. Herbilabs Labware specializes in certified high-purity reagents manufactured specifically for demanding research applications. Every product undergoes rigorous testing with full documentation provided through comprehensive certificates of analysis.

Our sterile reconstitution solutions deliver the purity essential for peptide research and sensitive biological assays. Manufactured in dedicated facilities with strict contamination controls, these solutions eliminate variables that compromise experimental outcomes. Whether you’re reconstituting lyophilized peptides or preparing cell culture media, starting with ultra-pure materials sets the foundation for reliable results. Access our detailed aseptic preparation checklist to ensure your handling techniques preserve reagent integrity throughout use. Explore the complete range of research-grade products and expert resources at Herbilabs to support your scientific excellence.
Frequently asked questions about high-purity reagents
What defines high-purity reagents in lab settings?
High-purity reagents typically contain 99.5% or greater of the specified compound with rigorously controlled impurity levels. These materials undergo comprehensive testing for contaminants like heavy metals, endotoxins, and residual solvents. Certificates of analysis document exact composition for each batch.
How often should reagent purity be verified during experiments?
Verify purity through certificates of analysis before starting any new project and when opening new reagent lots. For long-running studies, periodic testing every 3-6 months catches degradation that occurs during storage. Always retest if reagents show visible changes or produce unexpected results.
What are the risks of using low-purity reagents for peptide synthesis?
Low-purity reagents introduce competing side reactions that create deletion sequences and truncated peptides, dramatically reducing target yield. Contaminants can also modify amino acids during coupling, producing peptides with altered biological activity. These impurities often prove difficult to separate from desired products during purification.
Can high-purity reagents extend the shelf life of prepared solutions?
Yes, starting with ultra-pure reagents eliminates contaminants that catalyze degradation reactions in prepared solutions. However, shelf life still depends on proper storage conditions, sterile preparation technique, and the inherent stability of compounds in solution. High purity provides the best starting point but doesn’t eliminate other stability factors.
How do COAs help in selecting the right reagent batch?
Certificates of analysis reveal actual impurity levels and testing dates for specific lots, allowing you to choose batches with the lowest contamination for critical experiments. COAs also confirm that storage hasn’t degraded reagents by comparing current specifications against original manufacturing data. This batch-level detail prevents using compromised materials that meet general specifications but have elevated impurities in particular lots.
Recommended
- Aseptic Techniques During Reagent Preparation – A PRO Researcher’s 1# Checklist – Herbilabs Labware
- Understanding Reconstitution Solutions in 2025 Research Environments – Herbilabs Labware
- Reconstitution Solution 10x3ml – Sterile Solution in Premium Glass Vial – RUO – Herbilabs Labware
- Reconstitution Solution 10ml – Sterile Solution in Premium Glass Vial – RUO – Herbilabs Labware



