Inefficient consumables management quietly erodes research budgets, delays experiments, and creates compliance risks that compound over time. Budget losses up to 30% are attributable to poor inventory practices, including overstocking, expiry waste, and unplanned emergency orders. For laboratory managers and procurement specialists across European research institutions, these are not abstract risks but daily operational realities. This guide addresses each stage of the consumables workflow, from procurement planning and zoned storage to digital automation and waste reduction, providing step-by-step, evidence-backed strategies that translate directly into measurable cost savings, improved reliability, and stronger regulatory compliance.
Table of Contents
- Essential requirements for a lab consumables workflow
- Procurement planning and supplier selection steps
- Automating inventory tracking and storage with digital tools
- Waste management, expiry handling, and troubleshooting common failures
- Why traditional workflows fall short and what science-driven labs do differently
- Optimize your laboratory workflow with Herbilabs solutions
- Frequently asked questions
Key Takeaways
| Point | Details |
|---|---|
| Digital inventory cuts errors | Using digital tools and expiry alerts minimizes manual mistakes and reduces lost consumables. |
| Procurement planning saves budget | Centralized, strategic purchasing prevents waste and unlocks bulk discounts while avoiding overstock risks. |
| Diversify suppliers for resilience | Maintaining backup suppliers protects against delays and increases supply reliability. |
| FIFO and expiry tracking reduce waste | First-in, first-out management and expiry alerts can cut waste by up to 40% in laboratory settings. |
| Workflow audits improve outcomes | Regularly reviewing workflow steps and tools helps laboratories sustain efficiency and quality. |
Essential requirements for a lab consumables workflow
Before implementing any specific step, it is important to establish the foundational resources and structural prerequisites that make an efficient workflow possible. Attempting to optimize procurement or storage without these elements in place typically results in fragmented processes and recurring bottlenecks.
A well-designed consumables workflow rests on three interdependent pillars: organizational structure, physical infrastructure, and digital tools. On the organizational side, a core workflow includes procurement planning, quality assessment, inventory receipt, zoned storage, usage tracking, and waste management. Each stage requires defined roles and approval authority.
Three-level approval structure
Most European research institutions benefit from a three-level approval model:
- Application level: Individual researchers submit consumables requests with justification and project codes.
- Project review level: Principal investigators or department heads verify alignment with active projects and budgets.
- Procurement order level: A centralized procurement officer or team finalizes supplier selection and places orders.
This structure prevents unauthorized spending, supports audit trails, and reduces duplicate ordering across research groups.
Zoned storage infrastructure
Physical storage must be organized by temperature requirement and hazard classification. A functional zoned system typically includes ambient storage for general plasticware and dry reagents, refrigerated storage at 4°C for temperature-sensitive materials, ultra-low storage at minus 20°C or minus 80°C for biological reagents and standards, and a segregated hazardous materials zone with appropriate ventilation and containment. Reviewing lab consumables examples helps procurement teams map which items belong in each zone before designing storage layouts.

Digital tools and key consumables categories
A lab consumables comparison across product categories reveals that the most frequently managed items include syringes, pipette tips, parafilm, sample containers, filtration units, and reconstitution reagents. Each category carries distinct storage, expiry, and reorder requirements. Effective inventory management essentials include real-time stock dashboards, automated expiry alerts, and threshold-based auto-reorder triggers.
| Requirement | Manual approach | Digital approach |
|---|---|---|
| Stock visibility | Periodic manual counts | Real-time dashboard |
| Expiry management | Visual checks | Automated alerts |
| Reorder triggers | Reactive, ad hoc | Threshold-based automation |
| Compliance records | Paper logs | Audit-ready digital records |
Pro Tip: Set up multi-zoned storage with digital tracking from day one. Retrofitting these systems after a lab is operational is significantly more disruptive and costly than building them into initial setup.
Procurement planning and supplier selection steps
With the foundational infrastructure in place, procurement planning becomes the primary lever for controlling costs and ensuring supply continuity. Poorly planned procurement is one of the most common sources of both budget overruns and experimental delays in research labs.
Step-by-step procurement process
- Forecast demand based on active project timelines, historical usage data, and upcoming experimental schedules.
- Define quality benchmarks for each consumable category, including certification requirements, lot traceability, and compatibility with existing assays.
- Submit structured purchase requests through the three-level approval process described above.
- Evaluate supplier options using criteria such as lead time, pricing tiers, quality certifications, and regulatory compliance.
- Place orders with primary and backup suppliers, documenting lot numbers and expected delivery dates.
- Receive and inspect shipments against purchase orders, checking for damage, correct specifications, and expiry dates.
Centralized procurement at institutions such as the University of Innsbruck demonstrates that consolidating purchasing across departments yields measurable bulk discounts and reduces administrative time. However, centralized models can reduce flexibility for specialized or urgent requests.
| Factor | Centralized procurement | Decentralized procurement |
|---|---|---|
| Cost efficiency | Higher bulk discounts | Lower volume leverage |
| Flexibility | Lower for urgent needs | Higher for specialized items |
| Compliance oversight | Easier to standardize | Requires distributed controls |
| Administrative load | Concentrated in one team | Distributed across departments |
Bulk buying reduces per-unit costs but introduces risks. Balancing bulk buying with storage limits and diversifying suppliers are both critical for operational resilience. Overpurchasing ties up capital and increases expiry-related waste, particularly for reagents with short shelf lives.
For lab quality control purposes, procurement teams should require certificates of analysis for all certified lab supplies, particularly reconstitution reagents and biological standards. Reviewing consumables selection criteria before finalizing supplier agreements prevents costly compatibility issues downstream. Inventory management workflows should be integrated with procurement systems to provide automatic demand signals.
Pro Tip: Always maintain 2 to 3 backup suppliers for critical consumables. A single-supplier dependency creates significant vulnerability to regulatory delays, quality recalls, or production shortfalls.
Automating inventory tracking and storage with digital tools
After procurement, the most impactful operational upgrade a laboratory can make is transitioning from manual to digital inventory tracking. Manual systems, typically spreadsheets or paper logs, are inherently reactive and prone to human error, particularly in high-throughput research environments.
Manual versus digital workflow impact
The performance difference is substantial. Digital inventory using RFID and barcode systems enables an 84% reduction in inventory time, compressing checks that previously took 60 minutes down to approximately 10 minutes. This efficiency gain compounds across multiple inventory cycles per month.
Step-by-step digital implementation
- Audit current inventory to establish baseline stock levels, expiry dates, and storage locations.
- Select a platform that supports barcode, QR code, or RFID tagging based on your lab’s scale and budget.
- Label all consumables upon receipt, linking each item to its lot number, expiry date, and storage zone.
- Configure expiry alerts to notify procurement staff at defined intervals before critical items expire.
- Set reorder thresholds for each consumable category, triggering automatic purchase requests when stock drops below defined minimums.
- Integrate with cloud dashboards to give authorized staff real-time visibility across all storage zones.
| Metric | Manual workflow | Digital workflow |
|---|---|---|
| Inventory check time | 45 to 60 minutes | 8 to 10 minutes |
| Stock loss to expiry | Up to 5% per cycle | Less than 1% with FIFO |
| Data accuracy | 70 to 80% | Greater than 98% |
| Compliance readiness | Requires manual compilation | Audit-ready in real time |
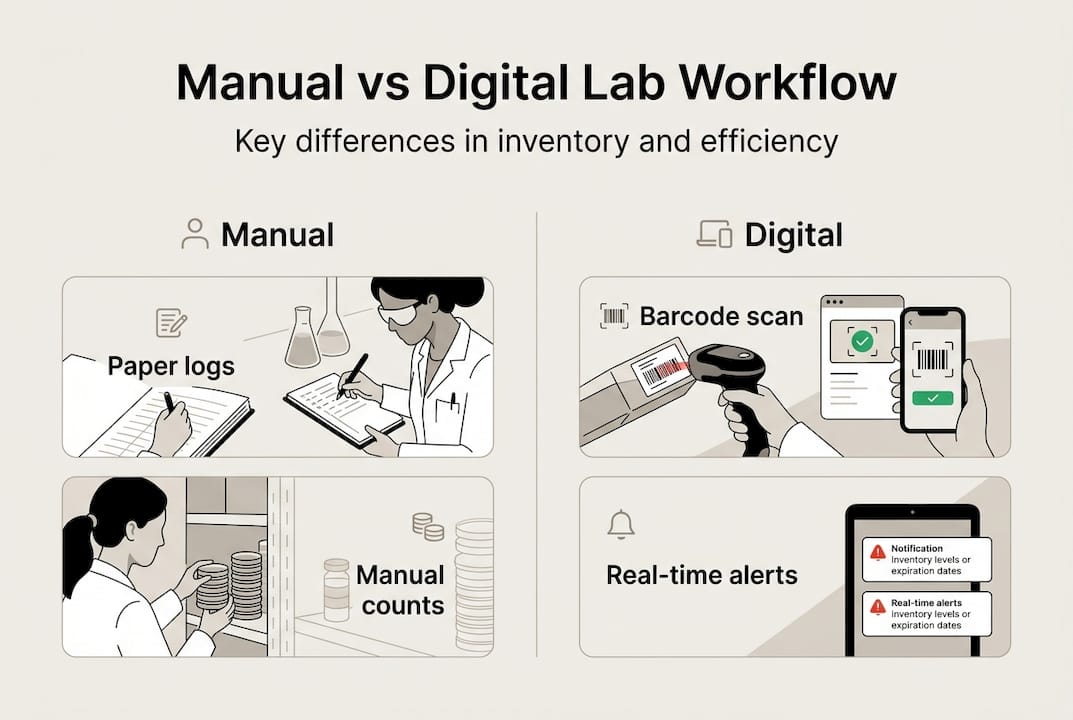
Digital systems support ISO 15189:2022 compliance by providing traceable, time-stamped records that align with risk management requirements for accredited laboratories. This is particularly relevant for European institutions operating under national accreditation frameworks.
For labs managing reconstitution reagents and sterile diluents, purity monitoring should be integrated into the digital tracking system to flag lot-specific quality deviations. Using lab inventory digital tools that combine stock management with quality data creates a single source of truth for procurement and compliance teams.
Pro Tip: Implement FIFO (first-in, first-out) protocols within your digital system. Configure the platform to automatically prioritize older stock for usage, which directly reduces expiry-based losses across all consumable categories.
Waste management, expiry handling, and troubleshooting common failures
Even well-designed procurement and tracking systems generate waste if expiry management and troubleshooting protocols are not actively maintained. Waste in laboratory consumables takes two primary forms: physical waste from expired or damaged materials, and financial waste from capital tied up in excess stock.
Overstock ties up 30 to 40% of new lab budgets, while a benchmark 5% stock loss to expiration is common in labs without structured FIFO protocols. Implementing FIFO consistently can reduce waste by up to 40%, representing a significant recovery of both budget and storage capacity.
“FIFO implementation reduces consumables waste by up to 40%, directly recovering budget and reducing the environmental footprint of laboratory operations.”
Top 5 common failures and fixes
- Overstocking: Driven by bulk-buying incentives without demand forecasting. Fix: Use historical usage data to set evidence-based reorder quantities.
- Expiry losses: Result from poor rotation and inadequate tracking. Fix: Enforce FIFO and configure automated expiry alerts at 90, 60, and 30 days.
- Lot-to-lot inconsistency: Lot inconsistency threatens reproducibility across experimental runs. Fix: Require lot-specific certificates of analysis and maintain lot records in your digital system.
- Assay failures from incompatible consumables: Incompatibilities cause assay failures that are difficult to diagnose. Fix: Validate new consumable lots against existing assay controls before full deployment.
- Single-supplier disruption: Regulatory delays or quality recalls from one supplier halt operations. Fix: Activate backup supplier agreements before primary stock falls critically low.
For lab safety compliance, expired reagents and hazardous consumables must be disposed of through approved waste streams, documented in the digital inventory system. Protocols for avoiding contamination during reconstitution and handling are equally important, as contaminated consumables generate both experimental failures and additional waste. Using inventory waste reduction platforms that track disposal events supports regulatory reporting requirements across European jurisdictions.
Pro Tip: Use data-driven forecasting tools to model expiry risk across your current stock. Many digital inventory platforms now include predictive analytics that flag items at high risk of expiration before losses occur.
Why traditional workflows fall short and what science-driven labs do differently
There is a persistent assumption in many research institutions that established manual routines, refined over years of practice, are reliable enough. The evidence does not support this view. Manual workflows are prone to discrepancies and waste, and they do not align with ISO 15189:2022 risk management or sustainability requirements that are increasingly mandatory across European accreditation frameworks.
Bulk buying is often treated as an unqualified win. In practice, it is only advantageous when storage capacity, expiry timelines, and demand forecasts are all accounted for simultaneously. Labs that bulk-buy without these constraints in place routinely generate the very waste they were trying to avoid.
Science-driven labs distinguish themselves by treating inventory data as a research resource. They run regular workflow audits, not just annual reviews, to identify systemic inefficiencies before they become budget crises. They also recognize that labware purity insights are part of the inventory story, not separate from it.
Labs that optimize workflows systematically can save up to 18% annually through reduced waste, better supplier terms, and fewer emergency orders. The shift from reactive to proactive management is not a technology problem. It is a process discipline problem that digital tools make significantly easier to solve.
Pro Tip: Schedule quarterly workflow audits that review procurement patterns, expiry data, and supplier performance together. Isolated reviews of each area miss the interactions between them that drive the most significant inefficiencies.
Optimize your laboratory workflow with Herbilabs solutions
Applying these workflow strategies requires not only the right processes but also reliable, high-quality consumables that perform consistently across experimental runs. Herbilabs supports laboratory managers and procurement specialists with resources designed to inform better purchasing decisions.

Explore the lab consumables comparison guide to evaluate product options against your workflow requirements, or review the high-purity reagents resource to understand how reagent quality directly affects assay reliability and reproducibility. For procurement teams seeking a trusted supply partner for bacteriostatic water, sterile diluents, and reconstitution solutions, Herbilabs solutions offer research-grade quality, rigorous quality control, and secure ordering tailored to European research institutions.
Frequently asked questions
What are the best practices for organizing laboratory consumables?
Zoned storage and multi-level approvals are established best practices: organize consumables by temperature requirement and hazard class, and route all procurement through a structured three-level approval process to maintain control and compliance.
How can digital inventory systems improve lab workflow?
Digital inventory checks drop from 60 to 10 minutes with barcode or RFID systems, while automated expiry alerts and real-time dashboards eliminate the reactive stock management that drives most consumables-related delays.
Why is supplier diversification important for laboratory consumables?
Diversifying suppliers is critical for operational resilience: maintaining 2 to 3 backup suppliers for critical items ensures that regulatory delays, quality recalls, or production shortfalls at one supplier do not halt laboratory operations.
How do laboratories minimize consumables waste and budget loss?
FIFO reduces waste by 40% when combined with expiry tracking and demand-based reorder quantities, preventing the overstocking that ties up 30 to 40% of first-year lab budgets in excess inventory.
What are common mistakes new labs make in consumables workflow?
Overstocking, lack of expiry tracking, single-supplier reliance, and regulatory delays are the most frequent workflow mistakes, each of which is preventable with structured procurement processes and digital inventory tools implemented from the outset.
Recommended
- Top lab consumables: key examples for efficient research
- Top 6 Lab Consumables Comparison Guide 2026
- Aseptic Techniques During Reagent Preparation – A PRO Researcher’s 1# Checklist – Herbilabs Labware
- Laboratory reagent handling: best practices for safety
- Manutenzione chimica industriale: guida alle best practice – Evoluzione Chimica
- Optimera arbetsflödet för industrimätning i svensk tillverkning



