TL;DR:
- Supply chain failures at any node can cause prolonged research delays and reagent shortages.
- Strategies like dual sourcing, risk-based inventory, and predictive analytics enhance lab supply resilience.
- Modern procurement tools and contingency planning are essential for managing temperature-sensitive and critical reagents.
A single delayed shipment of bacteriostatic water or a critical reagent can halt an ongoing peptide reconstitution study for weeks, forcing researchers to discard prepared samples and restart validation protocols from scratch. Laboratory supply chains involve sourcing raw materials, manufacturing, distribution, and last-mile delivery of reagents, consumables, and equipment, making them far more complex than standard procurement. Many laboratory managers underestimate this complexity until a stockout occurs at the worst possible moment. This article maps the core stages of the lab supply chain, identifies where failures concentrate, and outlines the strategies and technologies that high-performing research institutions use to maintain continuity.
Table of Contents
- Understanding the laboratory supply chain: Core stages and risks
- Key strategies for reducing supply chain disruptions
- Modern tools and technologies: From procurement platforms to AI
- Regulatory compliance and edge cases: What labs can’t afford to miss
- A fresh perspective: Why resilience tops cost savings in lab supply chains
- Next steps: Get reliable supply solutions for your lab
- Frequently asked questions
Key Takeaways
| Point | Details |
|---|---|
| Supply chain complexity | Lab supply chains involve more than ordering—they require careful coordination of procurement, storage, temperature control, and compliance. |
| Risk reduction tactics | Using dual sourcing, inventory segmentation, and predictive analytics greatly reduces the chance of disruptions. |
| Digital tools advantage | Modern procurement platforms and AI streamline sourcing, compliance, and fulfillment for labs. |
| Resilience vs. cost | Prioritizing reliability over short-term cost savings builds stronger, future-proof supply chains. |
Understanding the laboratory supply chain: Core stages and risks
The modern laboratory supply chain is not a simple ordering process. It is a multi-stage system where each node carries its own failure probability, and a breakdown at any point can cascade into research delays, compliance violations, or financial losses.
Key stages include procurement, inventory management, cold chain logistics for temperature-sensitive items like reagents, and regulatory compliance, all of which must function in coordination for uninterrupted laboratory operations. For bacteriostatic water and sterile diluents specifically, the stakes are elevated because these materials are subject to strict purity standards and contamination controls that add layers of complexity to every stage.
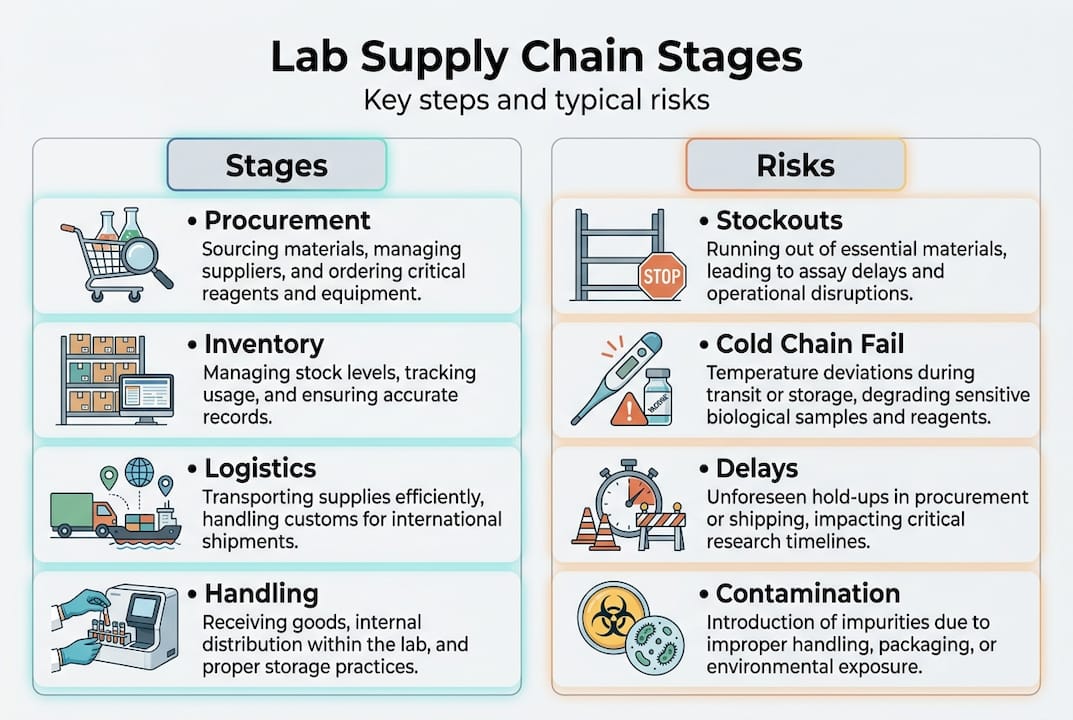
The following table maps the primary stages and their associated risk categories:
| Supply chain stage | Primary risk | Impact on reagent availability |
|---|---|---|
| Raw material sourcing | Single-source dependency | Immediate shortage |
| Manufacturing | Quality deviation, batch failure | Delayed or rejected stock |
| Warehousing | Temperature excursion, contamination | Product loss |
| Procurement | Lead time unpredictability | Stockouts |
| Cold chain logistics | Cold chain failure | Reagent degradation |
| Last-mile delivery | Customs delays, mishandling | Delayed access |
For research-grade materials, temperature sensitivity is a defining vulnerability. Reagents requiring refrigerated or frozen transport are exposed to cold chain risk at every handoff point, from the manufacturer’s loading dock to the laboratory receiving area. A single excursion above the validated temperature range can render an entire batch unsuitable for use.
Common risk areas that procurement specialists should monitor include:
- Single-source suppliers for validated reagents with no pre-qualified backup
- Cold chain failures during transit, particularly for international shipments
- Lead time unpredictability driven by raw material shortages or regulatory holds
- Tier-2 and tier-3 supplier disruptions that are invisible until they surface as shortages
- Regulatory non-compliance at the import stage, causing customs holds
Understanding the lab consumables workflow in detail is essential before attempting to build resilience, because you cannot protect a process you have not fully mapped. Similarly, knowing which key lab consumables carry the highest criticality score helps prioritize where to focus risk mitigation efforts first.
Key strategies for reducing supply chain disruptions
Understanding these risks, what practical strategies actually work to keep lab shelves stocked and research on track?
Core methodologies include mapping vulnerabilities using critical materials registers, dual sourcing for redundancy, risk-based inventory segmentation, predictive analytics for demand forecasting, and supplier diversification to mitigate disruptions. These are not theoretical frameworks. They are operational practices that distinguish labs with consistent throughput from those that experience recurring shortages.
Vulnerability mapping and critical materials registers are the foundation. A critical materials register catalogs every reagent and consumable by its supply risk score, which accounts for supplier concentration, lead time, regulatory sensitivity, and substitutability. Items like bacteriostatic water from a single validated manufacturer score high and require dedicated mitigation plans.
Dual sourcing addresses the single-supplier problem directly. By pre-qualifying a secondary supplier before a disruption occurs, labs avoid the scramble to validate an alternative under time pressure. This is especially important for sterile diluents and reconstitution solutions where any new supplier must meet the same purity and quality standards as the primary source.
Effective strategies for building supply chain resilience include:
- Maintain a critical materials register updated quarterly, with risk scores and mitigation owners assigned
- Implement dual sourcing for all single-source reagents and high-criticality consumables
- Apply inventory segmentation: hold higher safety stock for long-lead or single-source items
- Use predictive analytics to model demand based on research project pipelines, not just historical usage
- Establish regional backup suppliers to reduce cross-border logistics exposure
- Conduct lab consumables comparison reviews annually to identify emerging alternatives
Pro Tip: When building your critical materials register, assign each item a continuity score based on three factors: number of qualified suppliers, lead time in days, and regulatory substitutability. Items scoring high on all three require immediate dual-sourcing action.
Demand forecasting deserves particular attention. Most labs rely on historical consumption data, but this approach fails when research programs shift, new assays are introduced, or project funding changes. Integrating project pipeline data with inventory planning produces significantly more accurate forecasts and reduces both stockouts and excess inventory. Efficient material control systems provide the operational backbone for this kind of integrated planning across large research institutions.

The consumables workflow must also be reviewed when implementing these strategies, because workflow inefficiencies often mask underlying supply chain vulnerabilities that only become visible during a disruption.
Modern tools and technologies: From procurement platforms to AI
Once sound strategies are in place, what technology is reshaping how labs manage and secure their supply chains?
Procurement platforms and AI tools enable real-time comparison, fulfillment tracking, and diversification, reducing backorders and pricing volatility impacts. The shift from manual purchase orders to integrated digital platforms represents a meaningful operational upgrade for laboratories managing dozens of reagent lines simultaneously.
The following comparison illustrates how leading procurement platforms differ in their capabilities:
| Feature | Traditional procurement | Digital platform (e.g., Zageno) | AI-augmented system |
|---|---|---|---|
| Price comparison | Manual, periodic | Real-time, automated | Predictive, trend-adjusted |
| Stock visibility | Supplier-reported | Live inventory feeds | Risk-flagged alerts |
| Order automation | Manual requisition | Automated reorder triggers | AI-optimized timing |
| Compliance tracking | Manual SDS filing | Integrated SDS lookup | Automated regulatory alerts |
| Supplier diversification | Relationship-dependent | Multi-supplier catalog | AI-ranked alternatives |
For laboratory managers, the practical benefits are measurable. Automated reorder triggers eliminate the manual monitoring burden that typically falls on research staff who are not procurement specialists. Real-time stock visibility across multiple suppliers allows procurement teams to redirect orders within hours when a primary supplier reports a shortage.
Key technology applications that labs should prioritize include:
- Procurement platform integration with laboratory information management systems (LIMS) for seamless order triggering based on inventory thresholds
- AI-driven demand forecasting that incorporates project timelines, seasonal usage patterns, and supplier lead time variability
- Automated SDS (Safety Data Sheet) lookup to maintain regulatory compliance without manual document management
- Real-time fulfillment tracking with exception alerts for delayed or temperature-excursion-flagged shipments
- Supplier risk scoring dashboards that aggregate financial stability, delivery performance, and quality metrics
Pro Tip: When evaluating procurement platforms, prioritize those with direct API integration to your LIMS. Manual data entry between systems is where compliance gaps and ordering errors concentrate, and eliminating that interface removes a significant source of risk.
Maintaining consistent reagent standards becomes significantly easier when procurement technology enforces specification filters at the point of ordering, preventing the accidental purchase of lower-grade materials that could compromise assay validity.
Regulatory compliance and edge cases: What labs can’t afford to miss
While digital tools and strategies help, regulations and exceptional cases require their own solutions.
Edge cases include single-source reagents for validated methods, temperature-controlled materials prone to cold chain failures, custom chemicals with long lead times, tariff-induced cost increases, and tier-2 and tier-3 supplier disruptions causing 50% of issues. Each of these scenarios requires a specific contingency plan, not a generic response.
Regulatory compliance obligations that labs must address include:
- Import and export documentation: Reagents crossing international borders require accurate classification codes, certificates of analysis, and in some cases import licenses
- Storage temperature logs: Validated cold chain materials require continuous temperature monitoring records that are audit-ready
- Expiry and batch traceability: Regulatory frameworks in research institutions require full traceability from supplier batch to experimental use
- Controlled substance handling: Certain reagents require additional licensing and storage controls that must be reflected in procurement and inventory systems
“Pre-qualify secondary suppliers before disruptions occur, quantify financial exposure from tariff changes, use AI for SDS lookup and risk assessment, and maintain contingency plans including backup suppliers and alternate materials.” 5 Questions Every Lab Manager Should Ask
For tariff-induced cost increases, the recommended approach is to calculate the cost-per-test impact of a 15% to 25% tariff increase on key imported reagents, then model whether domestic alternatives or bulk purchasing agreements can offset that exposure. This is a financial planning exercise that procurement specialists should conduct annually, not reactively.
Proper reagent handling practices and aseptic techniques during reconstitution are also part of the compliance picture, because contamination events that originate from handling errors are often misattributed to supply chain failures, obscuring the true root cause. Robust lab quality control systems help distinguish between supplier-side and user-side quality events, which is essential for accurate supplier performance evaluation.
A fresh perspective: Why resilience tops cost savings in lab supply chains
The most persistent mistake in laboratory procurement is optimizing for unit cost rather than supply continuity. This is understandable, because procurement performance is often measured by cost savings, not by disruptions avoided.
The shift from just-in-time to just-in-case inventory reflects a hard-won lesson from recent years of supply volatility. Labs that maintained buffer stocks and dual-sourced their critical reagents in 2025 continued operating while others paused studies for weeks. The cost of holding additional safety stock is almost always lower than the cost of a halted experiment when researcher time, consumables already used, and sample loss are factored in.
The practical recommendation is to model supply chain decisions on long-term continuity value, not historical price. A supplier offering 10% lower unit cost but with a single-facility manufacturing footprint and no regional backup carries a risk premium that rarely appears in the purchase order but always appears in the incident report. Reviewing your consumables workflow through a resilience lens, rather than a cost lens, changes which decisions look optimal.
Next steps: Get reliable supply solutions for your lab
Ready to take the next step toward resilient and efficient supply in your lab?
Herbilabs provides research-grade bacteriostatic water, sterile diluents, and reconstitution solutions manufactured to strict purity standards, with quality control processes designed to meet the demands of scientific research institutions and universities. Our resources are built to help procurement specialists and laboratory managers make informed sourcing decisions.

Explore our practical guide to selecting research reagents to understand the quality criteria that matter most for peptide reconstitution and assay work. You can also review our consumables comparison resource to evaluate options across key performance and compliance parameters. Reliable supply starts with informed sourcing, and we are here to support that process.
Frequently asked questions
What are the most common causes of lab supply chain disruptions?
Single-source dependencies, cold chain breakdowns, and customs delays are leading causes, with tier-2 and tier-3 disruptions accounting for 50% of supply issues in research environments.
How can laboratories manage temperature-sensitive reagents safely?
Validated cold chain logistics with continuous temperature monitoring and scheduled delivery windows are essential, as cold chain logistics represent one of the highest-risk stages for reagent integrity.
What are some effective ways to forecast supply needs in a lab?
Integrating project pipeline data with historical usage records and applying predictive analytics for demand forecasting produces significantly more accurate supply plans than consumption history alone.
How should labs prepare for supplier or import tariff changes?
Labs should pre-qualify backup suppliers, calculate cost-per-test impacts of tariff increases, and diversify sourcing to reduce exposure, as tariff-induced cost increases on imports can significantly affect reagent budgets without advance planning.
Recommended
- Laboratory reagent handling: best practices for safety
- How to select laboratory reagents for reliable peptide research
- Understand laboratory reagent standards for reliable results
- Why certified lab supplies are essential for reliable research
- Prodotti chimici B2B: guida per gestioni ottimali 2026 – Evoluzione Chimica



